
The electrons in an atom are arranged in shells which are, on average, further and further from the nucleus, and which can only hold a certain number of electrons. This is, in part, because the distribution of electrons is not completely random. It is undeniable that atoms do behave as if they were spheres with a radius of 30–300 pm, that atomic size varies in a predictable and explicable manner across the periodic table and that this variation has important consequences for the chemistry of the elements.Ītomic radius tends to decrease on passing along a period of the periodic table from left to right, and to increase on descending a group. 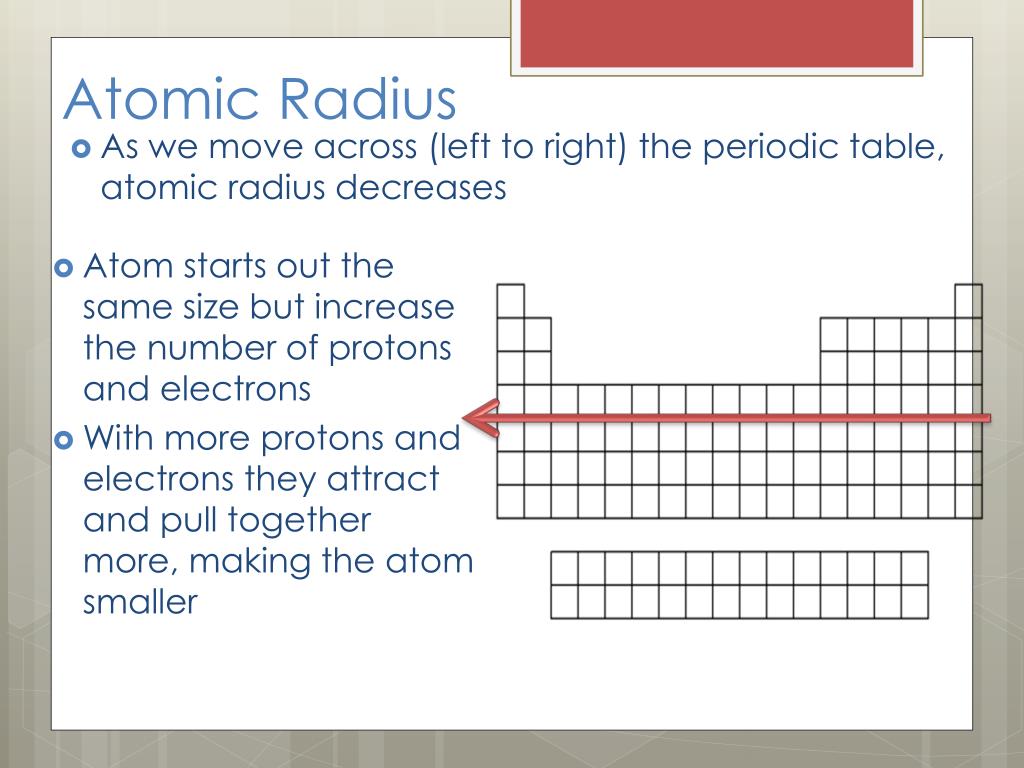
However the electrons do not have definite positions-although they are more likely to be in certain regions than others-and the electron cloud does not have a sharp edge.ĭespite (or maybe because of) these difficulties, many different attempts have been made to quantify the size of atoms (and ions), based both on experimental measurements and calculational methods. 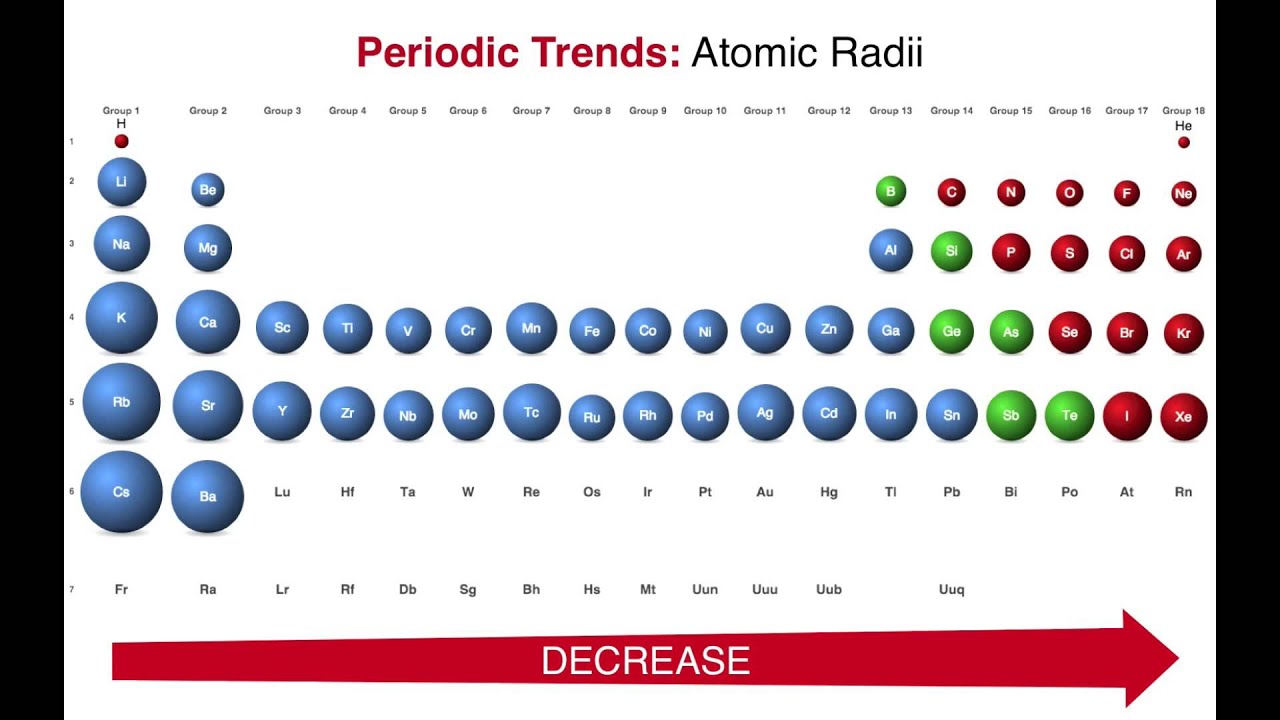
The atomic radius is determined entirely by the electrons: The size of the atomic nucleus is measured in femtometres, 100,000 times smaller than the cloud of electrons. In the latter case, which is the approach adopted here, it should also include ionic radius, as the distinction between covalent and ionic bonding is itself somewhat arbitrary. The term "atomic radius" itself is problematic: it may be restricted to the size of free atoms, or it may be used as a general term for the different measures of the size of atoms, both bound in molecules and free. The value assigned to the radius of a particular atom will always depend on the definition chosen for "atomic radius", and different definitions are more appropriate for different situations. Atomic radius patterns are observed throughout the periodic table.Atomic radius, and more generally the size of an atom, is not a precisely defined physical quantity, nor is it constant in all circumstances. The covalent radii of these molecules are often referred to as atomic radii. Nevertheless, it is possible for a vast majority of elements to form covalent molecules in which two like atoms are held together by a single covalent bond. Some are bound by covalent bonds in molecules, some are attracted to each other in ionic crystals, and others are held in metallic crystals. However, this idea is complicated by the fact that not all atoms are normally bound together in the same way. The atomic radius is one-half the distance between the nuclei of two atoms (just like a radius is half the diameter of a circle). This causes the electron to move closer to the nucleus, thus increasing the electron affinity from left to right across a period. Moving from left to right across a period, atoms become smaller as the forces of attraction become stronger. With a larger distance between the negatively-charged electron and the positively-charged nucleus, the force of attraction is relatively weaker. This means that an added electron is further away from the atom's nucleus compared with its position in the smaller atom. \( \newcommand\): Periodic Table showing Electron Affinity TrendĮlectron affinity generally decreases down a group of elements because each atom is larger than the atom above it (this is the atomic radius trend, discussed below).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
